Efficient Room Temperature, Ambient Pressure Liquid Ammonia Storage System
Case ID:
TEC2022-0082
Web Published:
5/14/2023
Executive Summary
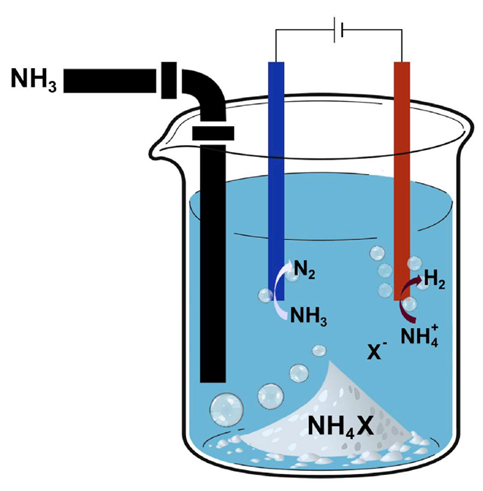
Liquid ammonia is current stored and transported in very large quantities in refrigerated units, railcars, tanker trucks and pressurized containers. While the majority of existing uses are as a nitrogen fertilizer for agricultural uses, increasing attention has been turned to ammonia as a green, carbon free energy storage medium for fueling combustion engines and fuel cells. However, the low boiling point (-33°C) and toxicity of ammonia presents challenges to safely storing and handling the material. MSU researchers have recently developed a new method of storing liquid ammonia under ambient conditions in a binary solution with a unique salt system. High concentrations of ammonia can be reversibly stored with this technology and ammonia vapors liberated from the solution in a controlled manner by the addition of mild heat.
Description of the Technology
This technology utilizes presents a simple and reversible way of storing ammonia at room temperature and ambient pressure through the formation of a solution which contains ammonia and the ammonium salts ammonium triflate (NH4OTf) or ammonium hexafluorophosphate (NH4PF6). Up to 2-3 equivalents of ammonia can be stored per each equivalent of salt to form a clear solution at 25°C. At this temperature, the weight fraction of ammonia is up to 25 wt.%. Increase temperature releases ammonia and the fraction of ammonia in the solution linearly decreases. Cyclic voltammetry and open circuit potential measurements show the solutions are also well suited for ammonia electrolysis.
Benefits
- Allows for the easy and safe storage of liquid ammonia
- Reversible
- Non flammable system
- Potential as electrolytes for ammonia splitting
Applications
- Bulk ammonia stationary or transport storage systems (chemical plants, agriculture, fuel, etc.)
- Safe storage medium for hydrogen used in fuel cells
- Electrolyte for direct conversion to H2 .
Patent Status
Patent pending
Licensing Rights
Full licensing rights available
References
Joule publication
Inventors
Dr. Chenjia Mi, Dr. Reza Ghazfar, Dr. Milton Smith, III, Dr. Thomas Hamann
TECH ID
TEC2022-0082
Patent Information:
| App Type |
Country |
Serial No. |
Patent No. |
File Date |
Issued Date |
Expire Date |
For Information, Contact:
Jon Debling
Technology Manager
Michigan State University
deblingj@msu.edu